Protein Structure Analysis and Visualization of Five Species
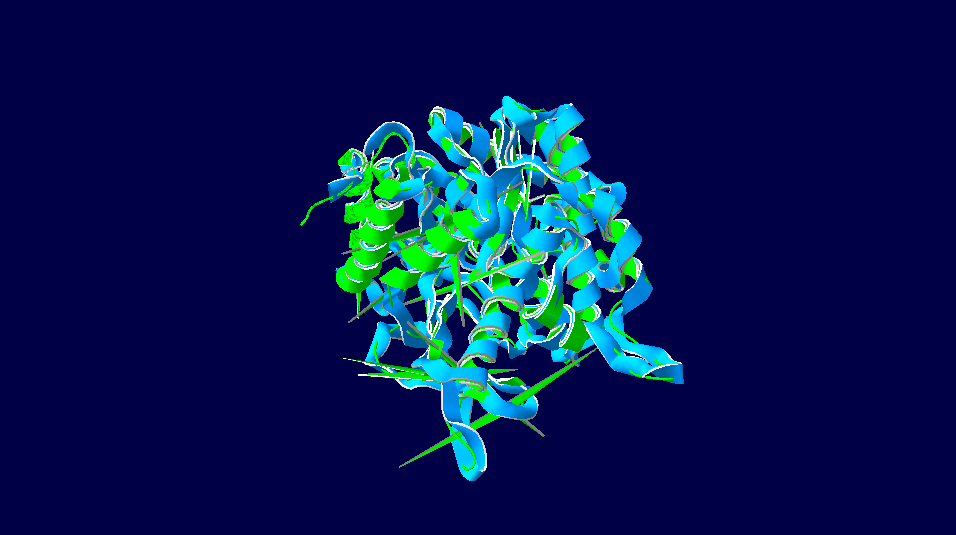
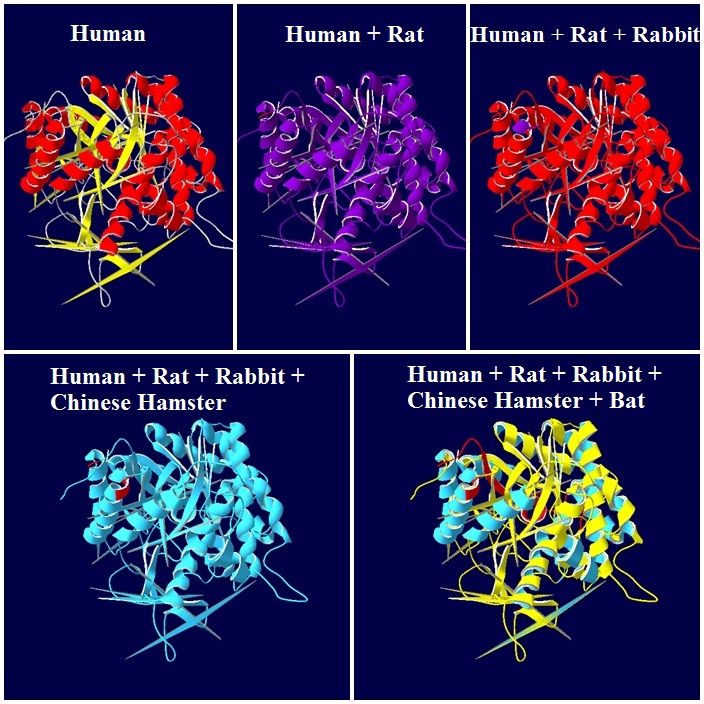
Homo sapiens versus Rattus norvegicus
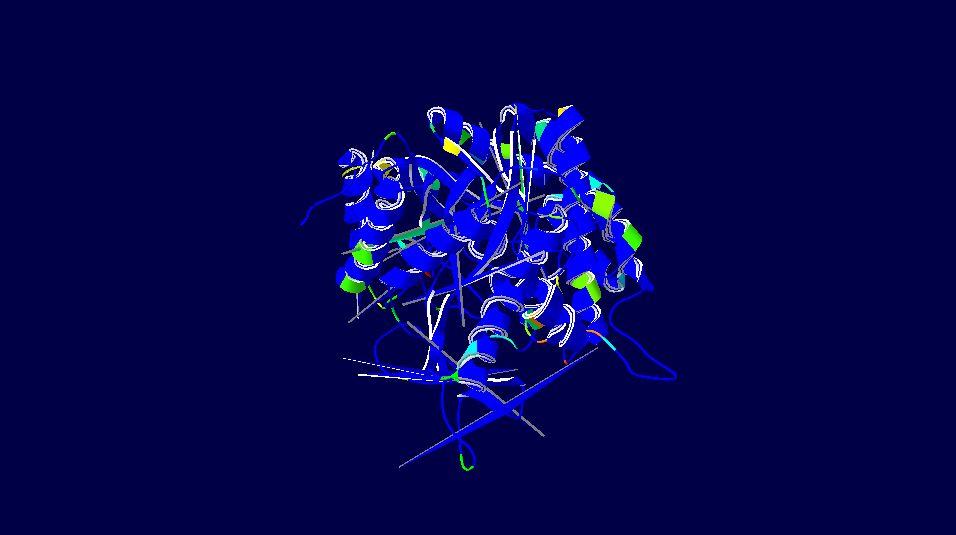
Homo sapiens versus Oryctolagus cuniculus
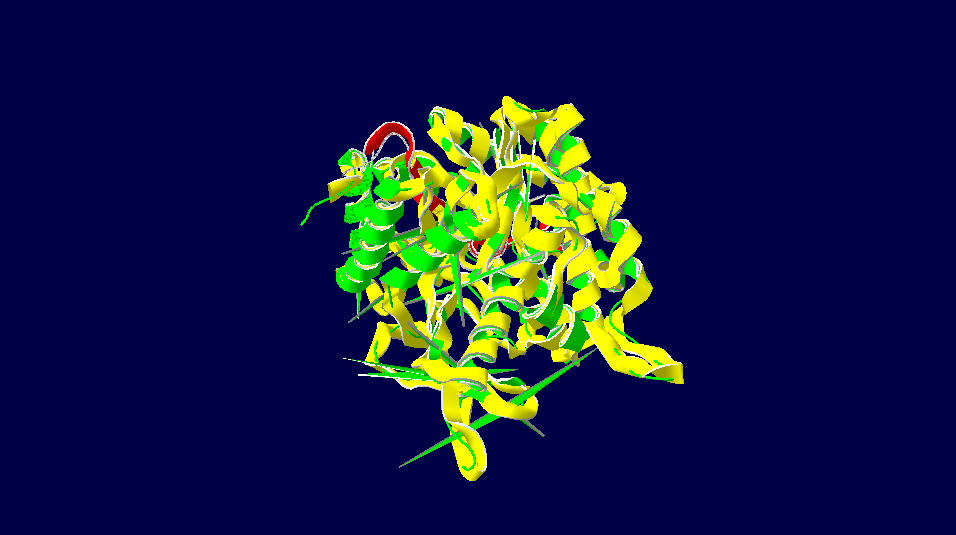
Homo sapiens versus Cricetulus griseus
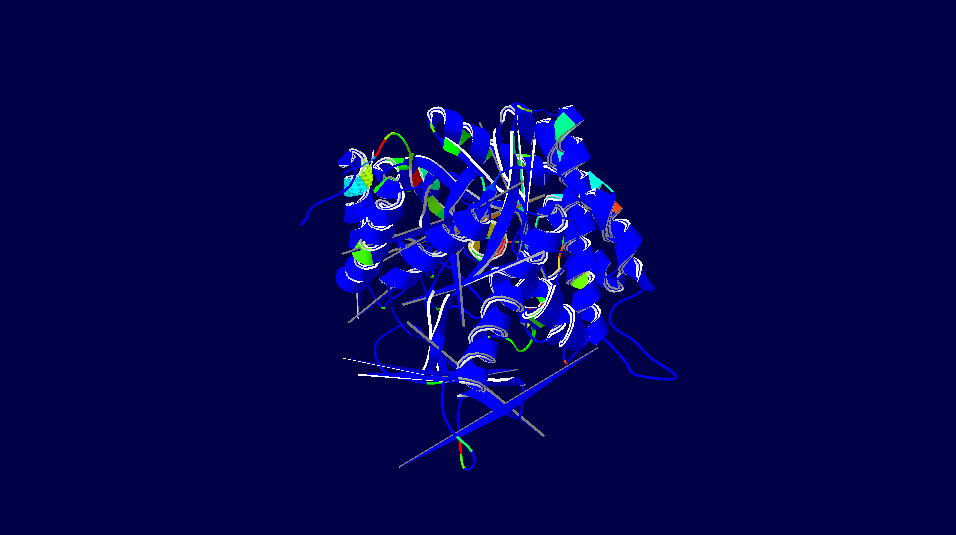
Homo sapiens versus Myotis davidii (David's myotis)
3D-Structure of 5 Species
Note: The colour schemes colour works on a cold → hot colour series where cold colours are the most conserved positions and hot colours are the most divergent. However, this fact only applies to the figures with alignment diversity.
Description on amino acids between Homo sapiens, Rattus norvegicus and Oryctolagus cuniculus
Analysis of the deduced amino acid sequence using bioinformatics revealed that mouse chromosomes shared extensive sequence homology with human LPH containing a conserved glycosy1 hydrolase family 1 motif important for regulating lactase intolerance. The functional gene is assigned to murine chromosome 1E3, which is different from the region of human chromosome 2 where the human lactase gene is located at 2q21.
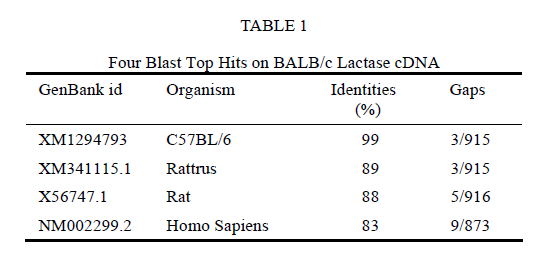
NCBI BLAST analyses indicated that the amplified DNA fragment was the homologue of human lactase gene. The comparison of this mouse lactase gene sequence with several other lactase sequences revealed high degrees of identities as shown in Table 1.
NCBI BLAST analyses indicated that the amplified DNA fragment was the homologue of human lactase gene. The comparison of this mouse lactase gene sequence with several other lactase sequences revealed high degrees of identities as shown in Table 1.
From Figure 9, we could find that the LPH protein had a transmembrane peptide segment at C-terminal. This indicated that the cloned cDNA encoding a 303 aa polypeptide chain contained a glycosyl hydrolase family 1 regain IYVTENGVS between amino acid residues 121 to 129.
The comparison of sequences in UniGene with proteins supported by a complete genome showed that the alignment between the cloned gene product and human lactase-phlorizin hydrolase precursor was 79.57%. The alignment between the cloned gene product and rat lactase-phlorizin hydrolase precursor was 87.78%, suggesting that the function of the cloned gene is similar to that of human LPH gene. Lloyd et al. reported that LPH is an integral microvillar membrane protein anchored by a transmembrane C-terminal peptide segment. It is synthesized on the rough endoplasmic reticulum which is then proteolytically converted to the final brush-border form.
General Description of Hydrogen Bonds and Disulphide Bonds
Lactase activity consists of wide range of substrate selectivity and their substrate can be found in several types of glycosides. Interaction of lactase with each of the substrate molecule can be investigated by using the monodeoxy derivatives of methyl β-lactoside. According to the journal, third hydroxyl group and sixth hydroxyl group were important for the recognition of the substrate by the enzyme. This has indicated third hydroxyl group and sixth hydroxyl group are involved in hydrogen bond forming and also acts as donor.
The pathway of folding protein into its final native configuration is still not fully defined. However, abundant of information has shown the protein folding are regulated by rapid interaction of hydrophobic residues in polypeptide chain, formation of secondary structure such as α-helices and β-sheets and finally formation of disulphide bond. Compact structural feature of protein that are stabilized by disulphide bridge.
Formation of disulphide bonds is an important step in the folding of lactase. For example, the LPHα profragment is rich in hydrophobic amino acids and contained 13 cysteine residues, while mature LPH (1061 amino acids) contain only 6 cysteine residues. This may show that these cysteine residues are involved in disulphide bond formation and their bond number different from different species.
Sources:
The pathway of folding protein into its final native configuration is still not fully defined. However, abundant of information has shown the protein folding are regulated by rapid interaction of hydrophobic residues in polypeptide chain, formation of secondary structure such as α-helices and β-sheets and finally formation of disulphide bond. Compact structural feature of protein that are stabilized by disulphide bridge.
Formation of disulphide bonds is an important step in the folding of lactase. For example, the LPHα profragment is rich in hydrophobic amino acids and contained 13 cysteine residues, while mature LPH (1061 amino acids) contain only 6 cysteine residues. This may show that these cysteine residues are involved in disulphide bond formation and their bond number different from different species.
Sources:
- Ralf Jacob, K. P., and
Hassan Y. Naim (2001). "The Prosequence of Human Lactase-Phlorizin
Hydrolase Modulates the Folding of the Mature
Enzyme" THE JOURNAL OF BIOLOGICAL CHEMISTRY 277(March 8): 8
- Ralf Jacob, N. J. B. a.
H. Y. N. "Folding of Human Intestinal Lactase-phlorizin Hydrolase"
from http://www.jbc.org/content/270/31/18678.full.