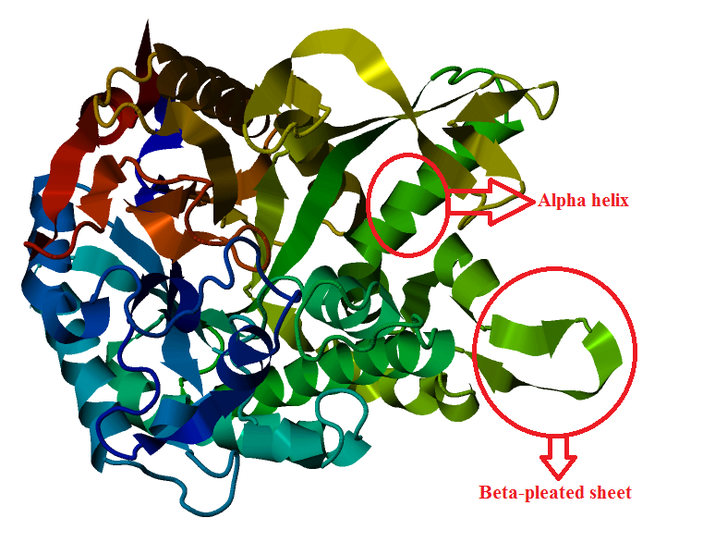
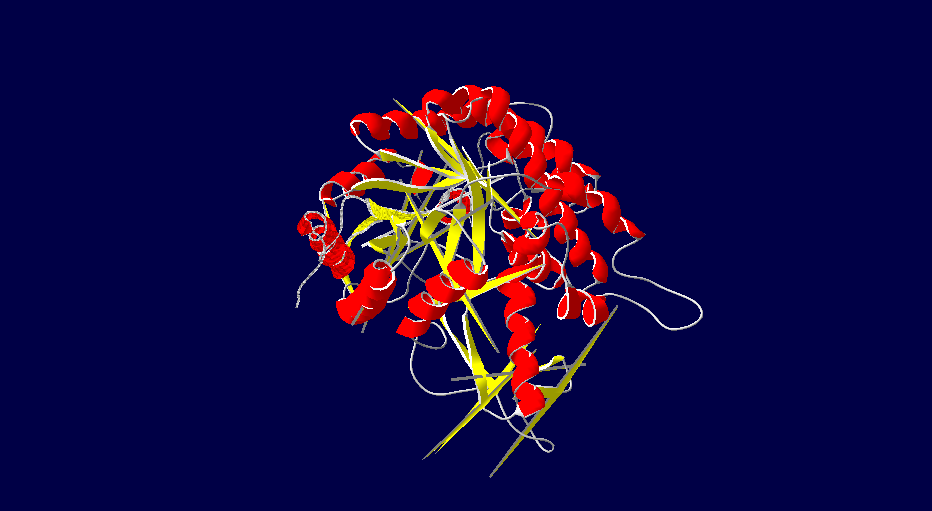
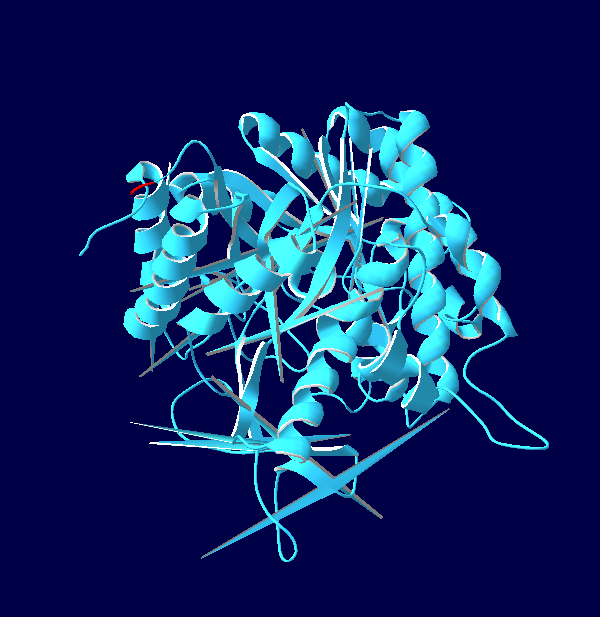
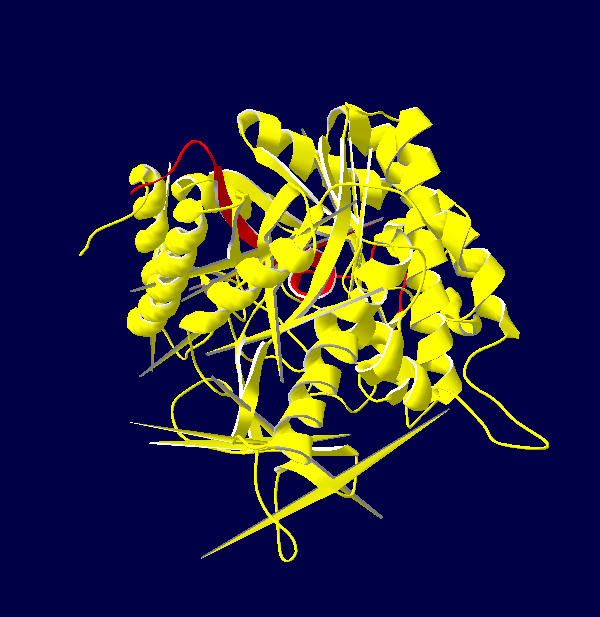
Protein Structure Modelling of Homo sapiens
Protein Structure Modelling of Rattus norvegicus (Rat)
Protein Structure Modelling of Oryctolagus cuniculus (Rabbit)
Protein Structure Modelling of Cricetulus griseus (Chinese Hamster)
Protein Structure Modelling of Myotis davidii (David's myotis) (Bat)
The 3D structures of Rattus norvegicus, Oryctolagus cuniculus, Cricetulus griseus and Myotis davidii are modeling using human structure as a template. From the structures that we modeled, it can be seen that all the five species shared the similar 3D structure. It might due to the selected species are belonging to the mammalia family. According to Hakulinen N. et al., 2003, the stabilizing factor for each protein was depend on the number of alpha-helix and beta-sheet in the protein secondary structure. The secondary structure of the 5 species comprises of equal amount of alpha-helix and beta-sheet chain. Large amount of beta-sheet structure confers the thermostability to the organisms. This indicates that longer beta strand is able to rigidify the protein and, thus make it more thermostable. On the other hand, it is believed that alpha-helix chain can confer the flexibility in the organisms. This is because high flexibility allows higher enzyme activity of the organisms.
Source: Hakulinen N. et al. (2003). Three-dimensional Structures of Thermophilic beta-1,4-xylanases from Chaetomium thermophilum and Nonomuraea flexuosa. Eur. J. Biochem, 270, 1399-1412.
Source: Hakulinen N. et al. (2003). Three-dimensional Structures of Thermophilic beta-1,4-xylanases from Chaetomium thermophilum and Nonomuraea flexuosa. Eur. J. Biochem, 270, 1399-1412.